PURPOSE
When formulating topical creams—or any preserved oil-and-water emulsion—there are varying philosophies on how best to incorporate preservatives. One traditional approach suggests that even oil-soluble preservatives should be added to the water phase of an oil-in-water (O/W) emulsion to ensure better contact with the aqueous phase and to avoid partitioning at the oil-water interface.¹ In contrast, another view recommends adding preservatives to their respective phases: water-based preservatives to the water phase and oil-based preservatives to the oil phase, prior to emulsification.² Additionally, the use of intermediate solvents to aid preservative incorporation is also common.³ The purpose of this study was to evaluate these techniques to determine whether the method of preservative incorporation affects its final distribution in a two-phase emulsion system.
METHOD
A simple emulsion system was developed for evaluation (Figure 1). The formulation consisted of an aqueous phase (~80%) and an oil phase (~20%, using isopropyl palmitate), with steareth-2 as the surfactant at either 2% or 6%. When testing the use of a co-solvent phase, 10% diethylene glycol monoethyl ether (DGME) was included in the aqueous phase. Six preservatives were evaluated: four antimicrobials (propylparaben, methylparaben, benzyl alcohol, phenoxyethanol) and two antioxidants (butylated hydroxyanisole [BHA] and butylated hydroxytoluene [BHT]). These were incorporated into different phases during the manufacturing process. A total of 10 emulsions were prepared:
Five emulsions with 6% surfactant, designed to form stable creams that were later separated into individual phases via centrifugation.
Five identical emulsions using only 2% surfactant, which allowed the emulsion to break spontaneously, enabling assessment of how quickly preservative partitioning occurs.
For all formulations, the aqueous and oil phases (each containing steareth-2) were heated separately to ~70°C before combining. Preservatives were added to specific phases as outlined below, followed by homogenization into a cream, and then cooled. The five emulsion types prepared were:
- No DGME; preservatives added to the aqueous phase
- No DGME; preservatives added to the oil phase
- DGME present; preservatives added to the aqueous phase
- DGME present; preservatives added to the oil phase
- DGME present; preservatives pre-dissolved in DGME and then added to the formed emulsion
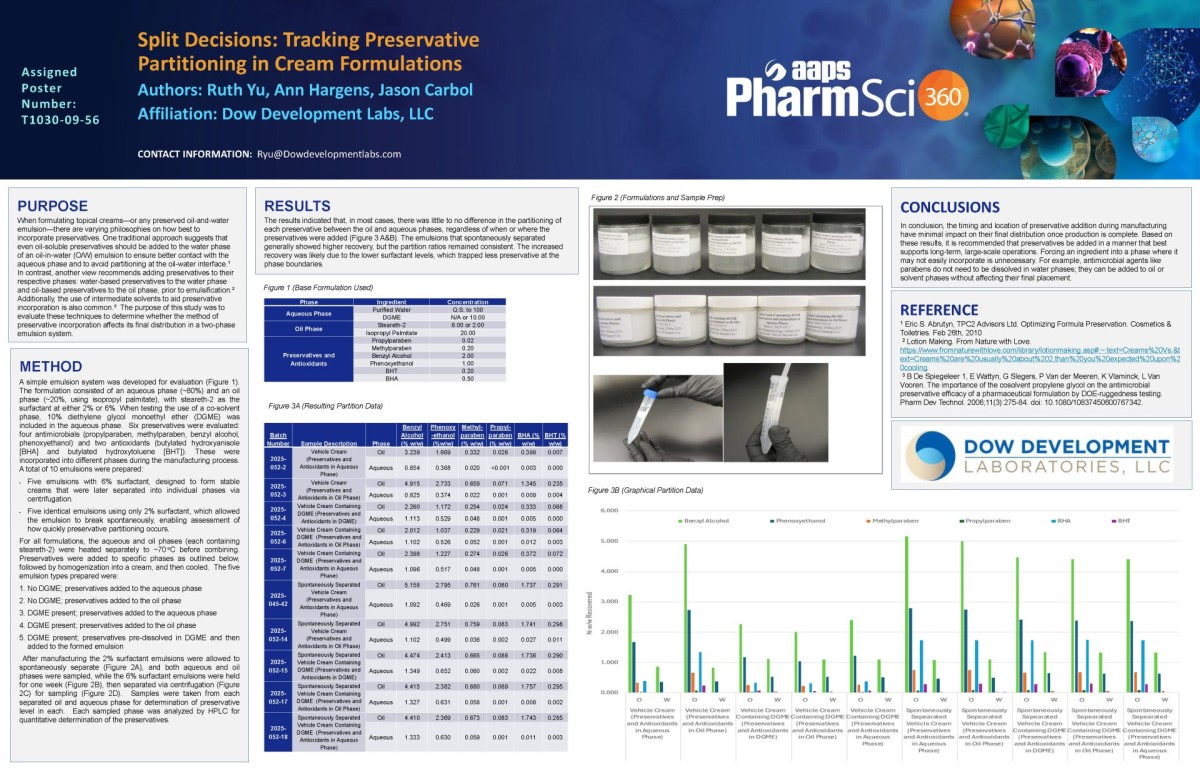
After manufacturing the 2% surfactant emulsions were allowed to spontaneously separate (Figure 2A), and both aqueous and oil phases were sampled, while the 6% surfactant emulsions were held for one week (Figure 2B), then separated via centrifugation (Figure 2C) for sampling (Figure 2D). Samples were taken from each separated oil and aqueous phase for determination of preservative level in each. Each sampled phase was analyzed by HPLC for quantitative determination of the preservatives.
RESULTS
The results indicated that, in most cases, there was little to no difference in the partitioning of each preservative between the oil and aqueous phases, regardless of when or where the preservatives were added (Figure 3 A&B). The emulsions that spontaneously separated generally showed higher recovery, but the partition ratios remained consistent. The increased recovery was likely due to the lower surfactant levels, which trapped less preservative at the phase boundaries.
CONCLUSIONS
In conclusion, the timing and location of preservative addition during manufacturing have minimal impact on their final distribution once production is complete. Based on these results, it is recommended that preservatives be added in a manner that best supports long-term, large-scale operations. Forcing an ingredient into a phase where it may not easily incorporate is unnecessary. For example, antimicrobial agents like parabens do not need to be dissolved in water phases; they can be added to oil or solvent phases without affecting their final placement.
REFERENCE
1 Eric S. Abrutyn, TPC2 Advisors Ltd. Optimizing Formula Preservation. Cosmetics & Toiletries. Feb 26th, 2010
2 Lotion Making. From Nature with Love. https://www.fromnaturewithlove.com/library/lotionmaking.asp#:~:text=Creams%20Vs.&text=Creams%20are%20usually%20about%202,than%20you%20expected%20upon%20cooling.
3B De Spiegeleer1, E Wattyn, G Slegers, P Van derMeeren, K Vlaminck, L Van Vooren. The importance of the cosolvent propylene glycol on the antimicrobial preservative efficacy of a pharmaceutical formulation by DOE-ruggedness testing. Pharm Dev Technol. 2006;11(3):275-84. doi: 10.1080/10837450600767342.\